- Blog
- About
- Contact
- Skyrim dragonborn dlc pc wont start
- Ocean of game xbox 360 games
- Outlook file extension
- Animated mouse cursor pack
- Nathan bronson
- Nonmem user guide
- Disk drill pro activation code
- Danzig misfits discography torrent
- Adobe audition cs6 mac trial
- Brunswick bristol ii pool table review
- Add pci to vm esxi 6-5
- Razorsql create table
- Miskatonic books discount code
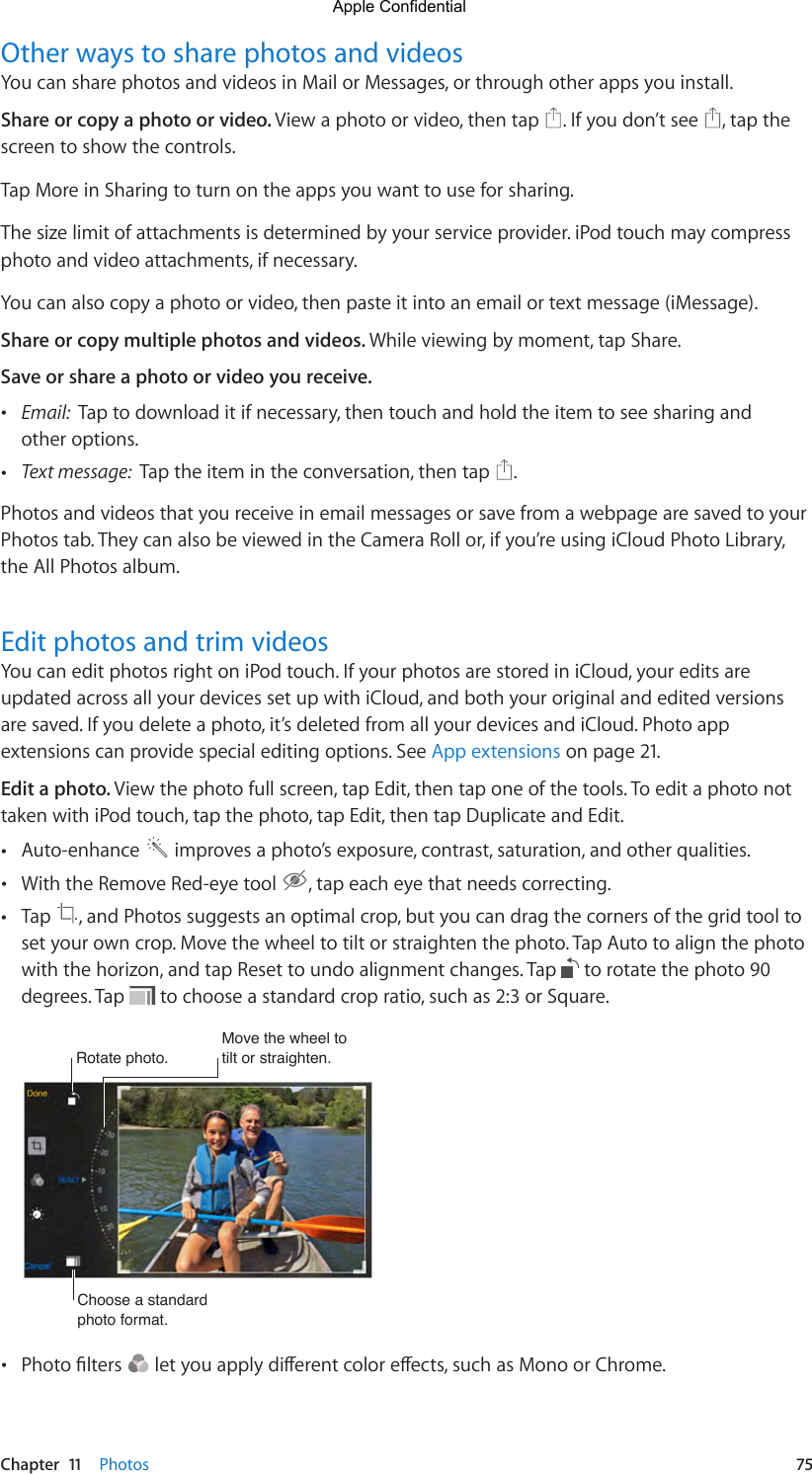
This requires knowledge of the structural PK model for the drug(s) being studied and formal consideration of how the population PK model is fitted to the data. Thus the intensive blood sampling that would make for ideal population PK modeling is not feasible, so the times at which the blood samples are taken must be chosen carefully so that information is maximised and the PK parameters can be estimated with reasonable precision. Therefore, there is an urgent need to characterise the concentration-time profile in vulnerable patient groups so that optimal dosing regimens can be determined.įor young children and pregnant women with uncomplicated malaria the invasiveness of taking many blood samples is not acceptable to the patient or parent. Previous studies have shown that anti-malarial pharmacokinetics for children and pregnant women can differ substantially from non-pregnant adults given the same dosing regimen. This drug concentration-time profile is determined by the pharmacokinetic (PK) properties of the drug. To treat malaria effectively, the dose and frequency of administration of the anti-malarial drug need to provide drug concentrations over time sufficient to kill all of the parasites in the body. This is especially true for young children and pregnant women, who carry much of the malaria burden and are at greatest risk of treatment failure. Most anti-malarial drugs were introduced with incomplete dose-finding studies, so the dosage, particularly in some important patient groups is too low. ACT involves treatment with two or more different anti-malarial drugs - a highly effective but short-lived artemisinin derivative, and a less potent but longer-lived partner drug(s). The World Health Organization recommends artemisinin-based combination therapy (ACT) as first line treatment for uncomplicated falciparum malaria in all malaria endemic areas. The proposed sampling designs in this paper are robust and efficient and should be considered in future PK studies of oral artesunate where only three or four blood samples can be collected. The optimal designs resulted in acceptable precision of the PK parameters.
#NONMEM USER GUIDE WINDOWS#
The derived optimal sampling windows were 17 to 29 minutes, 30 to 57 minutes, 2.5 to 3.7 hours and 5.8 to 6.6 hours for non-pregnant adults 16 to 29 minutes, 31 minutes to 1 hour, 2.0 to 3.4 hours and 5.5 to 6.6 hours for designs with non-pregnant adults and children and 35 to 59 minutes, 1.2 to 3.4 hours, 3.4 to 4.9 hours and 6.0 to 8.0 hours for pregnant women. The derived optimal designs were then evaluated via simulation-estimation.
#NONMEM USER GUIDE SOFTWARE#
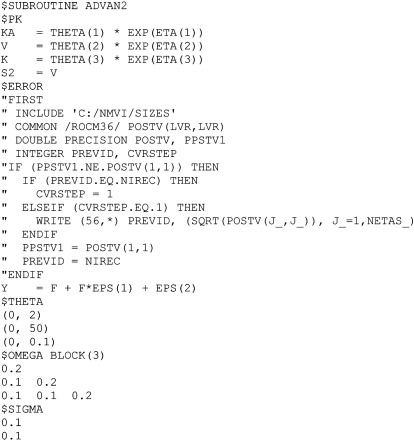
Optimal designs were derived using freely available software and were based on appropriate structural PK models from an analysis of data or the literature and key sampling constraints identified in a questionnaire sent to active malaria researchers (3-4 samples per patient, at least 15 minutes between samples). In this work optimal design methods were used to determine sampling designs for typical future population PK studies of dihydroartemisinin, the principal biologically active metabolite of oral artesunate. Optimal design methodology has been developed to determine blood sampling schedules that will yield precise parameter estimates within the practical constraints of sampling the study populations.
This could lead to imprecise estimates of the target PK parameters, and/or designs insufficient to estimate all of the parameters.

Currently, population pharmacokinetic (PK) studies of anti-malarial drugs are designed primarily by the logistical and ethical constraints of taking blood samples from patients, and the statistical models that are fitted to the data are not formally considered.
